

The future depends on rapidly detecting and treating contagious diseases, such as COVID-19, which is caused by the novel coronavirus.
Other diagnostic tests for COVID-19 may exceed 24 hours to produce results, making them impractical for testing at airports and borders. The VERI-Q PCR 316 utilizes real time PCR screening to analyze patients within one hour, meaning travellers may wait at the sample collection point for their test results. If the test comes back negative, the patient can be released; if the test is positive, they can be isolated and treated.
Many PCR screening systems rely on fluorescence, where targeted double-stranded DNA molecules are tagged with fluorescent dyes that emit light when exposed to a shorter wavelength input.2 These devices follow a similar schematic to fluorescence microscopes (Figure 1).
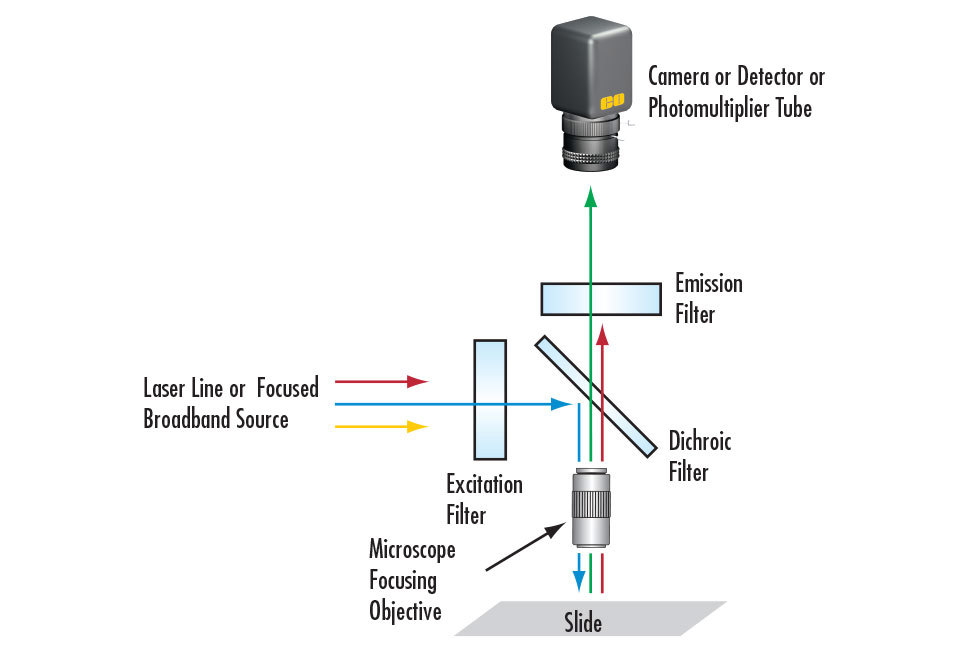
An excitation filter allows wavelengths from the illumination source that would excite the fluorescent sample to pass. These excitation wavelengths reflect off a dichroic filter and are focused onto the sample by lenses. Fluorescent emission from the sample transmits through the dichroic filter towards lenses that focus the light onto the system’s detectors. Measuring the amount of fluorescence lets the PCR screening system determine the presence of molecules that would indicate if a sample is COVID-19 positive.
Edmund Optics is proud to supply optical components for life-saving applications like MiCo BioMed’s VERI-Q PCR 316. This device, along with countless other optics-enabled technologies, are helping create a safer and healthier future.








or view regional numbers
QUOTE TOOL
enter stock numbers to begin
Copyright 2025 | Edmund Optics BV, De Maas 22B, 5684 PL Best, The Netherlands
California Consumer Privacy Acts (CCPA): Do Not Sell or Share My Personal Information
California Transparency in Supply Chains Act
This content may include material that has been generated or modified using artificial intelligence (AI).
The FUTURE Depends On Optics®